Flatiron’s Machine Learning Solution Benefits Cancer Patients and Big Pharma

Big Pharma Digs Deeps into their Wallets to Gain Access to Flatiron Health’s Machine Learning Technology, but will it be worth the price tag?
Clinical trials can be a bottleneck in the drug development process, particularly for anti-cancer therapies. Not only are clinical trials extremely expensive, but enrollment for cancer patients can be slow and difficult. The average cost of drug development was estimated to be more than $2.5 billion in 2015, with 60% of the costs comprised of clinical trials.[1] Flatiron Health vows to speed up the drug development by reducing the amount of extensive clinical trials currently required by the FDA (Figure 1).
By standardizing electronic health records (EHR) and using proprietary machine learning technology, Flatiron is helping to pave the way for data-driven precision medicine. The value of Flatiron’s EHR platform and its machine learning technology was attractive enough to be acquired by Roche Group in February 2018 for $1.9 billion.[2]

Figure 2: Flatiron uses real world endpoints to successfully predict results in Pfizer’s PALOMA-2 clinical trial for metastatic breast cancer.[6]
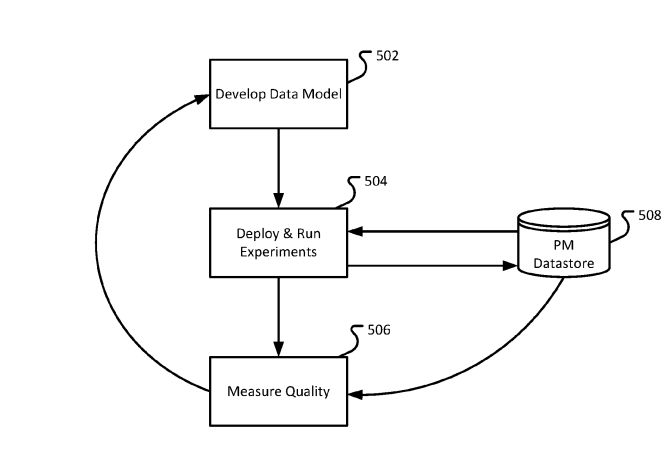
Flatiron’s future over the next few years looks bright. Flatiron’s patent pending abstraction process uses machine learning to improve and develop their data model.[7] This feedback loop allows their model to learn from new data coming in from their database and continue to learn, improve, and optimize (Figure 3). With the additional influx of capital from the $1.9 billion Roche deal, I would expect to see Flatiron make significant investments its scalability.
Figure 3: Data models are updated and learn from new data being extracted into the database. [7]
What can Flatiron Do to Maintain its Momentum?
Flatiron needs to continue to innovate and scale its reach over the next few years. To utilize machine learning accurately, the quality of the patient data set will be paramount. Expanding their data globally to increase variety and remove biases from the population sample set could make their data more reliable and widely applicable.
Flatiron will also need to reach out to regulatory agencies, such as the FDA, to engage regulators on how drug development and clinical trials may change with the introduction of machine learning over the next 5-10 years.
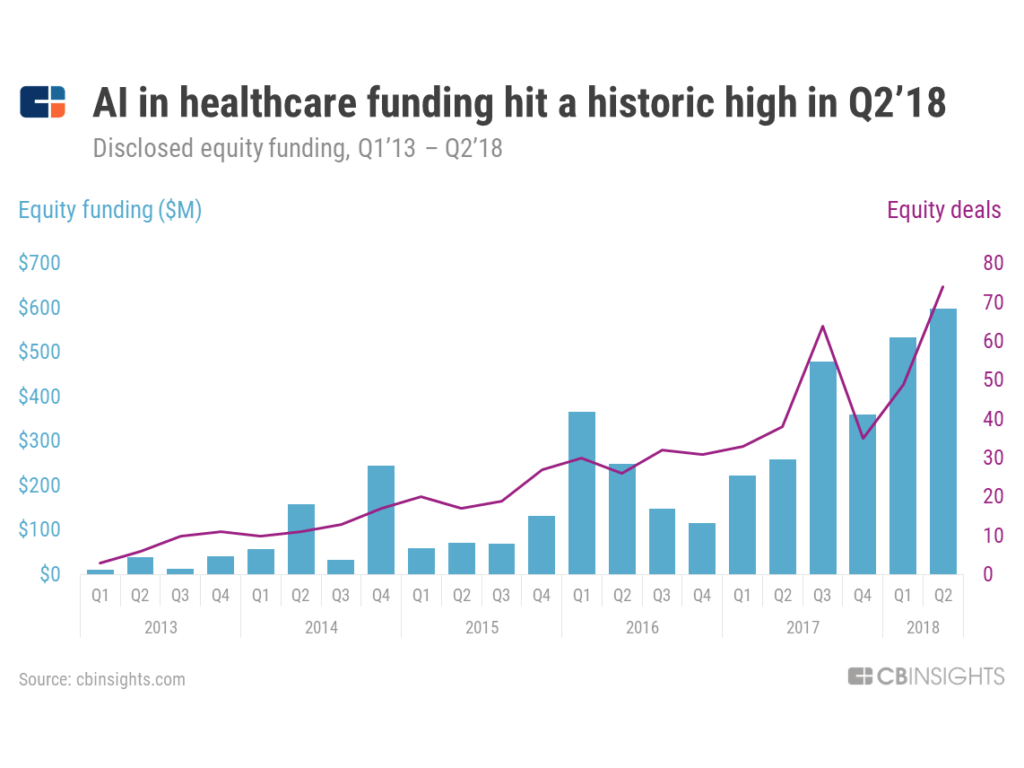
Flatiron will also need to keep an eye on their competition. The market for AI in health care is booming (Figure 4). To stay competitive, Flatiron should make significant investments in its technology and software platform. They should aim to increase the number of active cancer patients that are in this database over the next few years. Network effects will benefit the technology that Flatiron has to offer. As more patients, doctors, and pharmaceutical companies input data into the system, there will be increased benefits for all stakeholders. In this market, a significant increase in competition, especially in electronic health records, may hinder advancements due to a lack of standards for inputting medical data.
Figure 4: Healthcare funding for AI technologies has been increasing rapidly since 2013.[8]
Looking Forward
As the health care community eagerly waits for machine learning to be further integrated into general practice, there is, at this point, no approved AI-developed drugs.[9] Whether or not, the implications of machine learning on drug development is all hype or will have a real impact on the future of pharma is still to be decided. There is no question that big pharmaceutical companies have found these technologies attractive, but will these investments pay off?
(Word Count: 730)
[1] John Lechleiter, “Pharmaceutical Companies Sponsor Clinical Trials, But Don’t Directly Conduct Them,” Forbes, July 15, 2015, https://tinyurl.com/y7zqlj6q , accessed November 2018.
[2] Reenita Das, “The Flatiron Health Acquisition is a Shot in the Arm for Roche’s Oncology Real World Evidence Needs, ” Forbes, February 26, 2018, https://tinyurl.com/ya45eamq, accessed November 2018.
[3] “The Future of Clinical Trials: How AI & Big Tech Could Make Drug Development Cheaper, Faster, & More Effective,” CB Insights, August 7, 2018, https://tinyurl.com/y94arzh6, accessed November 2018.
[4] Erika Fry, “Tech’s Next Big Wave: Big Data Meets Biology,” Fortune, March 19, 2018, https://tinyurl.com/yauzffzh, accessed November 2018.
[5] Matthew Herper, “Flatiron Health, Purchased by Roche, Signs Three-Year Deal with Bristol Myers”, Forbes, May 2, 2018, https://tinyurl.com/yc7ya36k accessed November 2018.
[6] Flatiron Health, “Life Sciences: Accelerate research with the most advanced real-world evidence platform in oncology,” https://flatiron.com/real-world-evidence/, accessed November 2018.
[7] Gil Shklarski, “Extracting Facts from Unstructured Data,” US Patent Application: US 20170039341 A1. https://tinyurl.com/y7ye9mas, accessed November 2018.
[8] “The AI Industry Series: Top Healthcare AI Trends to Watch” CB Insights, https://tinyurl.com/y82nxafy, accessed November 2018.
[9] Nic Fleming, “How artificial intelligence is changing drug discovery,” Nature, May 30, 2018, https://www.nature.com/articles/d41586-018-05267-x, accessed November 2018.



I really like your closing remark about how big pharmaceutical companies have found these technologies attractive, but the real question is how much value can we possibly get from these. This is something that only time will tell. I found this particularly interesting because of the variety of industries that are using machine learning at the moment ( From reading the topics of our class’ blogs!). There is a possibility that many industries are trying to fit in these technologies into their businesses without a clear plan. This might be the case for Flatiron right now. However, given the importance of this field, healthcare in general and cancer in particular, even a slight advancement can add massive value to the world!